First passage time analysis of spatial mutation patterns reveals evolutionary dynamics of pre-existing resistance in colorectal cancer
 Sub-clonal mixing in human colorectal tumours
Sub-clonal mixing in human colorectal tumours
Abstract
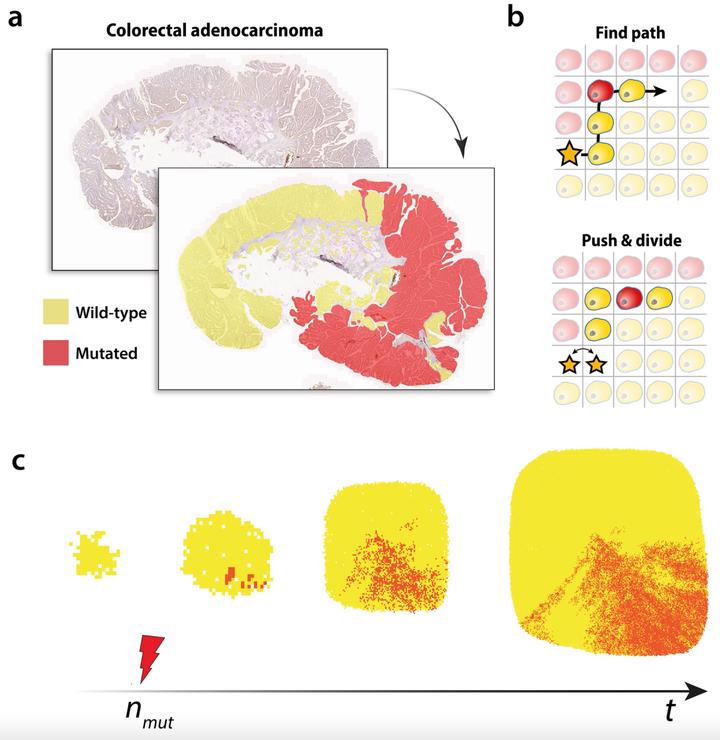
The signature of early cancer dynamics on the spatial arrangement of tumour cells is poorly understood, and yet could encode information about how sub-clones grew within the expanding tumour. Novel methods of quantifying spatial tumour data at the cellular scale are required to link evolutionary dynamics to the resulting spatial architecture of the tumour. Here, we propose a framework using first passage times of random walks to quantify the complex spatial patterns of tumour cell population mixing. First, using a simple model of cell mixing we demonstrate how first passage time statistics can distinguish between different pattern structures. We then apply our method to simulated patterns of mutated and non-mutated tumour cell population mixing, generated using an agent-based model of expanding tumours, to explore how first passage times reflect mutant cell replicative advantage, time of emergence and strength of cell pushing. Finally, we explore applications to experimentally measured human colorectal cancer, and estimate parameters of early sub-clonal dynamics using our spatial computational model. We infer a wide range of sub-clonal dynamics, with mutant cell division rates varying between 1 and 4 times the rate of non-mutated cells across our sample set. Some mutated sub-clones emerged after as few as 100 non-mutant cell divisions, and others only after 50,000 divisions. The majority were consistent with boundary driven growth or short-range cell pushing. By analysing multiple sub-sampled regions in a small number of samples, we explore how the distribution of inferred dynamics could inform about the initial mutational event. Our results demonstrate the efficacy of first passage time analysis as a new methodology in spatial analysis of solid tumour tissue, and suggest that patterns of sub-clonal mixing can provide insights into early cancer dynamics.